Back in 2020, the height of the COVID-19 pandemic brought increased attention to mental health and its related disorders. Mental health issues were seen on the rise, whether due to pandemic-related life changes, or infection with the virus itself.
Backing this data was a scientific brief on its Global Burden of Disease study, where the World Health Organization (WHO) estimated that the pandemic led to a 27.6% increase in cases of major depressive disorder (MDD) and a 25.6% increase in anxiety disorders (AD) worldwide in 2020 (World Health Organization, 2022).
But what’s been done about it? How has scientific research delved into understanding mental health interventions since this rise in 2020? Well, one core area that is increasingly being recognized as a potential factor in mental health is the link between our gut microbiome and mental health.
We now know that the gut microbiome, a collection of microorganisms (such as bacteria, viruses, and fungi) that reside in our digestive tract, plays a crucial role not only in digestion but also in various aspects of our physical and mental health.
In this research review and scientific guide, we’ll explore key pieces of research into the link between our gut microbiome and mental health, as well as the potential for diet-based interventions, or the use of probiotics and other interventions to improve mental health outcomes.
Background
In the USA, the 2021 State of Mental Health Report indicated that from January to September 2020, 315,220 people took the organization’s online screening test for anxiety—which was a 93% increase over 2019’s total number of screenings (Reinert et al., 2021). Additionally, during the pandemic, 4 in 10 U.S. adults reported symptoms of anxiety or depressive disorders, which was up from 1/10 who reported these symptoms from January to June 2019. Clearly, the pandemic has been a driving factor in increased mental health issues.
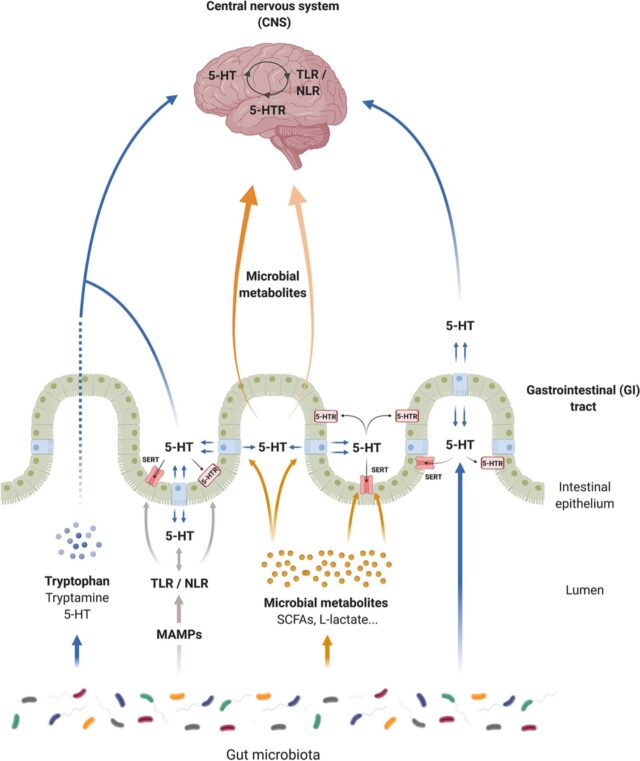
Recent studies show that the gut microbiome plays an important role in the gut-brain axis and, in turn, mental health. One of the major hormones influencing mental health is the neurotransmitter serotonin (Lin et al., 2014). Serotonin plays a range of roles in the body, including influencing mood, and a lack of it may lead to symptoms of depression and anxiety (Serotonin, 2022).
More than 90% of serotonin in the body is synthesized in the gut and gut microbiota play important roles in its regulation (Yano et al., 2015). Metabolites produced by gut microbes stimulate host gut enterochromaffin cells to produce serotonin, some of which enters circulation and travels to the brain to participate in the gut-brain axis (Layunta et al., 2021; Yano et al., 2015).
The gut-brain axis involves bidirectional communication between the central nervous system and the gut and facilitates the development and function of the immune, metabolic, and nervous systems (Layunta et al., 2021; Morais et al., 2021).
Direct evidence of the association between gut-brain communication and the microbiome comes mostly from rodent models, making it difficult to link the human gut microbiome to mental health. However, some recent studies shed light on this topic.
What do we already know about the gut-brain connection in simple terms?
To put the above background into layman’s terms, the gut microbiome, which consists of trillions of microorganisms living in our digestive system, plays a vital role in regulating serotonin levels in our body.
Serotonin is a neurotransmitter that influences our mood and lack of it can lead to mental illness, mental health decline and symptoms of depression and anxiety. This connection between the gut and brain function is known as the gut-brain axis and involves communication between the central nervous system and the gut to regulate various bodily functions.
Previous studies have shown that:
- The gut microbiome can produce important metabolites that influence serotonin levels
- Changes in the gut microbiome and gut bacteria can affect mental health and behavior in rodents
- The gut-brain axis is bidirectional, meaning our gut health can also impact our mental health
Possible interactions between diet, the gut microbiota, and mental health
Before we look at what the Bear et al research found, it’s important to understand how the gut microbiome can be influenced by external factors such as diet.
Even though it was earlier thought that the gut microbiota consisted of 500-1000 species, a recent large scale study estimates that the human gut microbiome consists of at least 1,800 genera and 15,000–36,000 species of bacteria.
These bacteria interact with the food we eat, and as such, different types of diets can affect the composition of our gut microbiome. For instance, a diet high in fat and sugar has been linked to reduced microbial diversity in the gut, which has been associated with various health issues (Lin et al., 2014).
On the other hand, a largely plant-based diet rich in fiber promotes the growth of beneficial bacterial species in the gut, leading to a more diverse and healthy microbiome composition.
With this in mind, we are collectively responsible for aiding digestion and other bodily functions.
But when we make poor or unhealthy dietary choices, it can lead to imbalances in our gut microbiome. These imbalances can impact a number of human diseases, from inflammation of the bowel in irritable bowel syndrome (IBS) and Celiac disease to the affect the production of important metabolites, like serotonin, leading to a potential decline in mental health.
This brings us to the question: can we use diet as an intervention for mental health issues? Let’s take a closer look at the research.
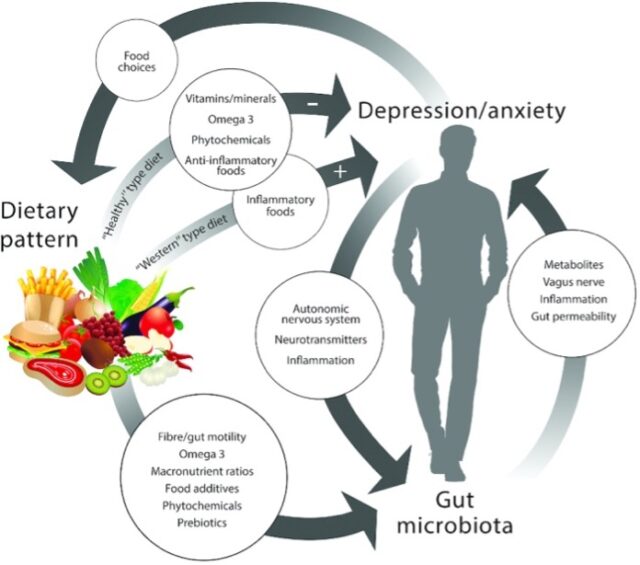
Bear et al, 2020
The Flemish Gut Flora Project, a large study of the gut microbiome in 1,054 individuals, examined how microbial community composition correlates with quality of life and depression symptoms (Valles-Colomer et al., 2019). This work found that the bacterial genera Faecalibacterium and Coprococcus, which are consistently correlated with positive health outcomes, are also associated with higher quality of life. When confounding factors were taken into account, depletion of Coprococcus, along with Dialister, was significantly associated with depression. Both Faecalibacterium and Coprococcus produce butyrate, a short chain fatty acid (SCFA) which has known positive effects directly on the gut, such as regulation of glucose metabolism and intestinal barrier integrity (Caspani et al., 2019). Administration of acetate, butyrate, and propionate, the three most abundant SCFAs, as well as sodium butyrate, improved symptoms of depression in mice, suggesting that bacterial SCFAs may play key roles in depression (Caspani et al., 2019). Another study of 5,959 genotyped individuals with matched gut metagenomic data found a possible causal association of Klebsiella and Morganella with depression, and further found increases of Morganella in 181 individuals who later developed depression (Qin et al., 2022). Both of these bacterial genera are gram-negative pathogens that induce inflammation, which may influence mood.
How the gut microbiota influence mood
Now that we know there are possible microbial links with mental health, how can we put this information to use, especially in the context of the COVID-19 pandemic? An easily modifiable solution is diet. It is well known that diet modulates the gut microbiota, to the point of causing community fluctuations after consumption of a single meal (David et al., 2013).
High-fiber foods enrich beneficial microbes which break fiber down into metabolites such as SCFAs (Makki et al., 2018). Therefore, consumption of a high-fiber diet may enrich beneficial, SCFA-producing bacteria such as Faecalibacterium and Coprococcus and reduce detrimental gram-negative pathogens, such as Klebsiella and Morganella.
In fact, both gut microbiota and diet have been linked to depression and anxiety (Bear et al., 2020).
Studies directly linking diet to depression are plagued with study design and interpretation difficulties, as well as conflicting results, making it difficult to establish a solid relationship. However, there is direct evidence linking changes in the microbiome to emotional behaviors associated with anxiety and depression, and since the microbiome is directly impacted by diet, this may explain the link between food and mental health (Bear et al., 2020).
Certain foods that benefit the microbiome, like omega-3 fatty acids, fiber, magnesium, vitamin D, and fermented foods, are also associated with improved mood. Although it is difficult to discern whether these foods are truly associated with depression, we can see that they directly alter the composition of the microbiome and enrich for beneficial taxa, suggesting that an overall healthy diet, like the Mediterranean diet, may improve depression and anxiety through modulation of the gut microbiome.
More research is needed to solidify and fully understand this link, but, in the meantime, it’s always beneficial to start incorporating microbiome-friendly foods and generally improve your diet and, in turn, your overall health.
Outcomes & Closing Thoughts
When we look to the future, it is clear that the gut microbiome and its relationship to mental health will continue to be an important area of research. The potential for using diet as a tool to improve mental health through modulation of the microbiome is exciting, but more research is needed before making any concrete recommendations or conclusions.
Overall, this emerging field highlights the interconnectedness between our body and what we consume, and the important role our gut microbiome plays in maintaining optimal health. By being mindful of our dietary choices and promoting a healthy, diverse microbiome, we can potentially influence not only our physical well-being but also our mental and emotional well-being.
At CosmosID, we provide a state-of-the-art microbiome sequencing and metagenomics platform to help researchers and healthcare providers better understand the impact of the microbiome on human health.
With our advanced technology and sequencing services, we are committed to advancing the field of gut microbiome research and providing valuable insights into this complex relationship between diet, gut microbes, and mental health.
Together, we can continue to uncover the mysteries of the microbiome and its role in improving overall human health. Contact us today.
References
Bear, T. L. K., Dalziel, J. E., Coad, J., Roy, N. C., Butts, C. A., & Gopal, P. K. (2020). The role of the gut microbiota in dietary interventions for depression and anxiety. Advances in Nutrition, 11(4), 890–907. https://doi.org/10.1093/advances/nmaa016
Caspani, G., Kennedy, S., Foster, J. A., & Swann, J. (2019). Gut microbial metabolites in depression: Understanding the biochemical mechanisms. Microbial Cell, 6(10), 454–481. https://doi.org/10.15698/mic2019.10.693
David, L. A., Maurice, C. F., Carmody, R. N., Gootenberg, D. B., Button, J. E., Wolfe, B. E., Ling, A. V, Devlin, A. S., Varma, Y., Fischbach, M. A., Biddinger, S. B., Dutton, R. J., & Turnbaugh, P. J. (2013). Diet rapidly and reproducibly alters the human gut microbiome. Nature. https://doi.org/10.1038/nature12820
Layunta, E., Buey, B., Mesonero, J. E., & Latorre, E. (2021). Crosstalk Between Intestinal Serotonergic System and Pattern Recognition Receptors on the Microbiota–Gut–Brain Axis. Frontiers in Endocrinology, 12(November), 1–24. https://doi.org/10.3389/fendo.2021.748254
Lin, S. H., Lee, L. T., & Yang, Y. K. (2014). Serotonin and mental disorders: A concise review on molecular neuroimaging evidence. Clinical Psychopharmacology and Neuroscience, 12(3), 196–202. https://doi.org/10.9758/cpn.2014.12.3.196
Makki, K., Deehan, E. C., Walter, J., & Bäckhed, F. (2018). The Impact of Dietary Fiber on Gut Microbiota in Host Health and Disease. Cell Host and Microbe, 23(6), 705–715. https://doi.org/10.1016/j.chom.2018.05.012
Morais, L. H., Schreiber, H. L., & Mazmanian, S. K. (2021). The gut microbiota–brain axis in behaviour and brain disorders. Nature Reviews Microbiology, 19(4), 241–255. https://doi.org/10.1038/s41579-020-00460-0
Qin, Y., Havulinna, A. S., Liu, Y., Jousilahti, P., Ritchie, S. C., Tokolyi, A., Sanders, J. G., Valsta, L., Brożyńska, M., Zhu, Q., Tripathi, A., Vázquez-Baeza, Y., Loomba, R., Cheng, S., Jain, M., Niiranen, T., Lahti, L., Knight, R., Salomaa, V., … Méric, G. (2022). Combined effects of host genetics and diet on human gut microbiota and incident disease in a single population cohort. Nature Genetics, 54(2), 134–142. https://doi.org/10.1038/s41588-021-00991-z
Reinert, M., Fritze, D., & Nguyen, T. (2021). The State of Mental Health in America in 2022.
Serotonin. (2022). Cleveland Clinic Website.
Valles-Colomer, M., Falony, G., Darzi, Y., Tigchelaar, E. F., Wang, J., Tito, R. Y., Schiweck, C., Kurilshikov, A., Joossens, M., Wijmenga, C., Claes, S., Van Oudenhove, L., Zhernakova, A., Vieira-Silva, S., & Raes, J. (2019). The neuroactive potential of the human gut microbiota in quality of life and depression. Nature Microbiology, 4(4), 623–632. https://doi.org/10.1038/s41564-018-0337-x
World Health Organization. (2022). Mental Health and COVID-19 : Early evidence of the pandemic’s impact. 2(March), 1–11.
Yano, J. M., Yu, K., Donaldson, G. P., Shastri, G. G., Ann, P., Ma, L., Nagler, C. R., Ismagilov, R. F., Mazmanian, S. K., & Hsiao, E. Y. (2015). Indigenous Bacteria from the Gut Microbiota Regulate Host Serotonin Biosynthesis. Cell, 161(2), 264–276. https://doi.org/10.1016/j.cell.2015.02.047



